How the winners of the Nobel Prize in Chemistry have transformed research and saved lives
The Nobel Prize in Chemistry 2018 has been awarded to three researchers for their work on “harnessing the power of evolution” to create compounds that are of benefit to humanity. One half of the nine million Swedish kronor (£770,686) prize will go to the American Frances Arnold from the California Institute of Technology, US. The other half will go jointly to the American George Smith from the University of Missouri, US, and the Brit Gregory Winter from the MRC lab in Cambridge, UK.


Marcos Alcocer , University of Nottingham
The Nobel Prize in Chemistry 2018 has been awarded to three researchers for their work on “harnessing the power of evolution” to create compounds that are of benefit to humanity. One half of the nine million Swedish kronor (£770,686) prize will go to the American Frances Arnold from the California Institute of Technology, US. The other half will go jointly to the American George Smith from the University of Missouri, US, and the Brit Gregory Winter from the MRC lab in Cambridge , UK.
We don’t run ads or share your data. If you value independent content and real privacy, support us by sharing.
Their work centres on techniques of “directed evolution” – a method which imitates natural selection. This can help to create new powerful proteins that achieve specific tasks.
The method is now widely used in the production of new synthetic drugs, such as recombinant antibodies , to process or produce biofuels and medical treatments. By engineering new molecules, the 2018 Nobel laureates have – according to the Royal Swedish Academy of Sciences – “taken control of evolution and used it for purposes that bring the greatest benefit to humankind”.
Useful mutations
One of the most crucial characteristics of evolving organisms – life as we know it – is the ability to replicate and mutate. All organisms can make copies of their genes and undergo changes that are passed to their progeny. In other words, we are totally dependent of evolving chemicals.

Arnold’s work focused on the directed evolution of enzymes – proteins that accelerate chemical reactions. Because they are so useful, scientists had long tried to create enzymes with desired properties artificially, but with little success.
Arnold – who is the fifth to join an important group of women to win the Nobel Prize in Chemistry – instead developed a method to produce mutations in the genes that produced certain enzymes in order to select the best ones. Different mutations will produce slightly different versions of the enzyme in each cell so, over time, one can select the one which works the best for a specific task.
Arnold’s discovery was hugely important – creating a completely new way to design and produce pharmaceuticals and renewable fuels for a greener transport sector.
Phage revolution
Smith and Winter also managed to use evolution to the advantage of humankind by developing a technique called phage display .
Smith first made the groundbreaking discovery of how “bacteriophages” – viruses that infect bacteria – work. Using standard DNA technology, Winter’s group then used the bacteriophages to evolve new proteins. Essentially, Winter used the phage technology in order to engineer new “antibodies” in the bacteria – large proteins that are used by the immune system to fight harmful bacteria and viruses. After many rounds of mutation and selection, artificial chemical evolution can select for the best antibody to fight a certain infection.
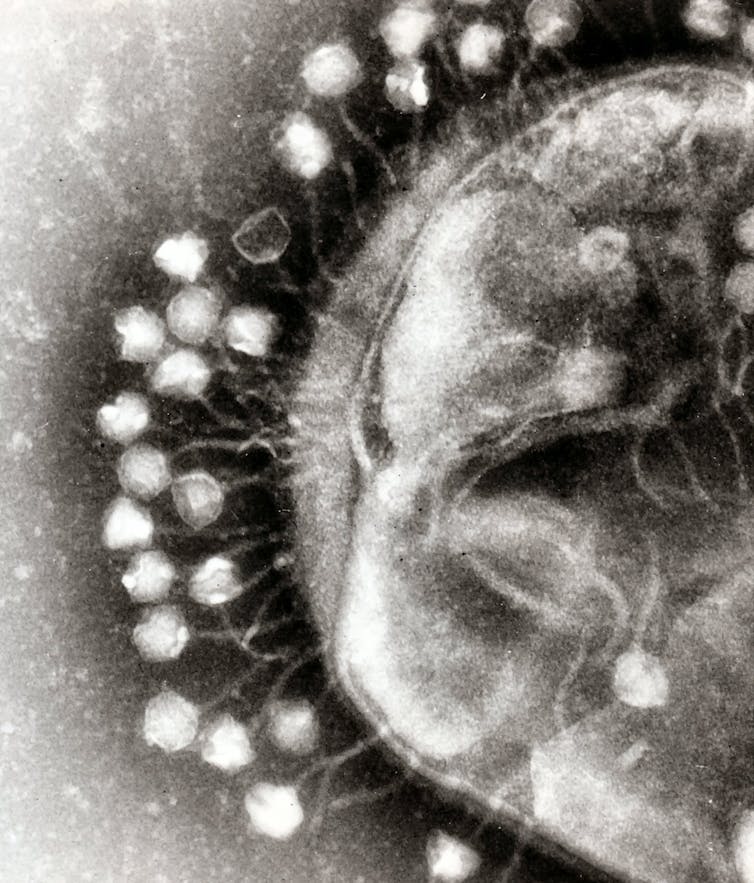
Winter was one of the first to produce functional mammalian antibodies or part of them in bacteria. Crucially, this moved the whole experimental area from being totally dependent on experimenting with animals to a complete new world of exact biology that could be performed in a simple Petri dish. It is therefore fair to say that one of the great contributions of their research was to reduce the number of animals used in the lab.
Winter has set up important commercial antibody producing facilities at Cambridge, producing drugs that can tackle devastating autoimmune diseases and metastatic cancer. Clearly his entrepreneurial vision, ability to surround himself by extremely competent people and openness to new techniques such as phage display makes him a worthy winner of this year’s Nobel Prize in Chemistry.
The work by all three recipients was largely carried out in the 1990s. Nowadays, scientists including myself base most of our research on synthetic libraries of proteins and enzymes, so it has truly transformed the entire field of protein engineering research as well as saving the lives of both animals and people. And this is only the start – we may see many more cures come from research involving these techniques in the future.
Marcos Alcocer , Associate Professor in Biochemistry, University of Nottingham
This article is republished from The Conversation under a Creative Commons license. Read the original article .
